Ozone hole one of the biggest ever – but don’t worry
- PostedPublished 29 November 2015
The Antarctic ozone hole reached 28.2 million square kilometres in October this year, the third largest observed after 2000 and 2006, according to NASA data.
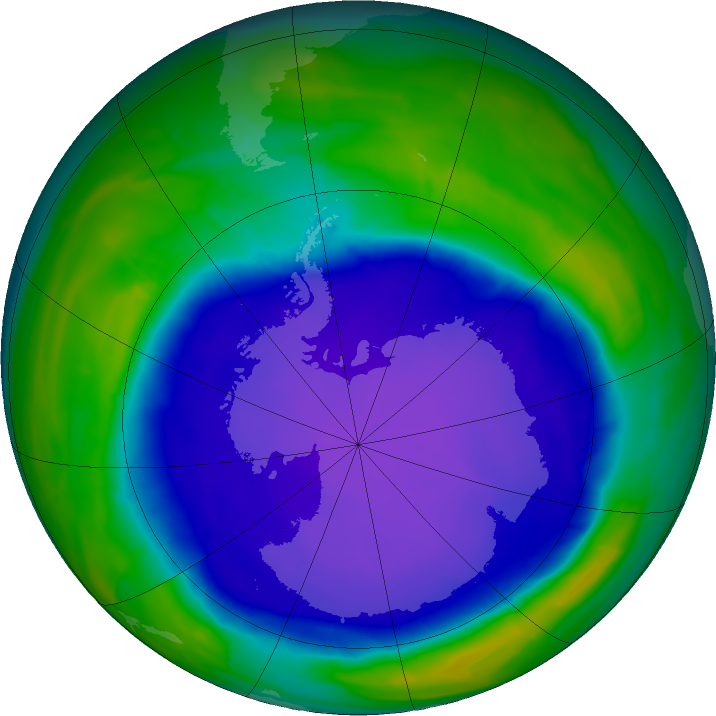
However rather than being a result of huge ozone depletion in the last 12 months, the World Meteorological Organisation (WMO) says we are not seeing a reversal of the good work achieved under the Montreal Protocol, but the effect of unusually cold stratospheric conditions.
National Oceanic and Atmospheric Administration (NOAA) instruments attached to balloons measured almost 100 per cent ozone depletion between 14 and 19 kilometres above the Earth’s surface at South Pole Station.
Usually measurements taken here during the final stages of usual September ozone decline end at around 95 per cent by October 1, but this year the depletion continued for an extra fortnight to cause the almost 100 per cent depletion reading, according to NOAA researcher Bryan Johnson.
WMO Atmospheric and Environment Research Division senior scientist Geir Braathen said that while the results are a reminder that the ozone problem remains and of the need to keep an eye on it, “there is no reason for undue alarm”.
“As a result of the broad compliance with the Protocol and industry’s development of ‘ozone-friendly’ substitutes for the now-controlled chemicals, the total global accumulation of ozone depleting substances has slowed and begun to decrease,” he said.
“Substantial recovery of the ozone layer is expected by the middle of the 21st century, although over the Antarctic it will be later – probably around 2070.”
NASA says the minimum ozone layer thickness recorded this year was 101 Dobson units on October 4, compared with between 250 and 350 Dobson units during the 1960s.
The Antarctic ozone hole was first discovered in the 1980s and initially blamed on air travel before scientists realised the link between chlorine and bromine in man-made products such as refrigerants with ozone depletion.
HFCs linked to ozone depletion: NASA
NASA has discovered that HFCs such as R134a do have an impact on ozone depletion after all, although it is small and indirect.
This news could strengthen the case for a phasedown of HFCs under the Montreal Protocol, which has so successfully reduced the use of ozone-depleting substances such as CFCs and HCFCs on a world-wide scale that the ozone layer is expected to have mostly recovered by the middle of this century.
Unlike CFCs and HCFCs, HFCs do not contain the chlorine that destroys ozone, but the fact they trap heat in the atmosphere speeds up the stratospheric chemical reactions that deplete ozone. And because warm air rises, ozone-poor air is lifted and further dilutes the ozone layer at tropical latitudes.
A NASA computer model predicted that HFCs will contribute to a global ozone reduction of 0.035 per cent by 2050.
NASA Goddard Space Flight Center research scientist Margaret Hurwitz explained that although this may seem like a small number, it is 100 times larger than previous estimates.
“Now that we understand the impacts of these particular greenhouse gases on the ozone layer, our next step is to study how these chemicals contribute to other changes in Earth’s climate and the environment,” she said.
In a nutshell, reducing HFC emissions will speed up the ozone layer’s recovery.
Suspected R134a manufacturing link to mystery ODS increase
You may remember that last year we reported that scientists were baffled by unexpected and previously unidentified ozone-depleting substances (ODS) in the atmosphere, considered the most significant increase in ODS since the Montreal Protocol was introduced.
Scientists now think that HCFC-133a, used in the manufacturing of R134a, had been leaking during the production process because atmospheric concentrations of the substance in the Northern Hemisphere rose rapidly between 2000 and 2013, then dropped suddenly by early 2015.
Martin Vollmer of the Swiss Federal Laboratories for Materials Science and Technology reckons the abrupt change signals human influence over the emissions and linked this to the fact that ozone-depleting HFC-133a is used in the mass production of ozone-friendly R134a.
“That’s most likely the biggest source of HCFC-133a currently,” he said. “But it’s not known how much leaks out. We expected HCFC-133a to grow rapidly in the atmosphere because of the enormous and growing global demand and production of HFC-134a, but we found the opposite. One of our speculations is that there are one, two or three factories that have been grossly emitted HCFC-133a but were cleaned up.”
- CategoriesIn SightGlass
- Tagsozone, SightGlass News Issue 4